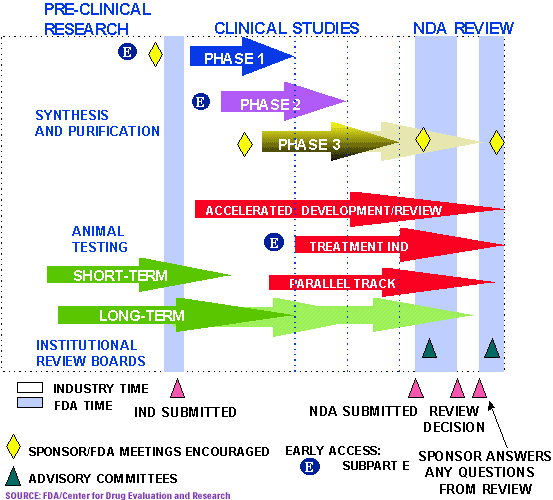
If the FDA gives the green light, the "investigative" drug will then enter three phases of clinical trials:
- Phase 1 uses 20-80 healthy volunteers to establish a drug's safety and profile. (about 1 year)
- Phase 2 employs 100-300 patient volunteers to assess the drug's effectiveness. (about 2 years)
- Phase 3 involves 1000-3000 patients in clinics and hospitals who are monitored carefully to determine effectiveness and identify adverse reactions. (about 3 years)
The research-based pharmaceutical industry currently invests some US$12.6 billion a year in new drug development. Historically, the drug development figure doubles every five years.
See also: How Drugs are Developed and Approved